A large emerald with a mass of 378.24 grams was recently discovered in a mine. If the density of the emerald is 2.76 grams over centimeters cubed, what is the volume? Round to the nearest hundredth when necessary and only enter numerical values, which can include a decimal point.
@kittybasil
do u have any idea on wat the answer is??
137.04
im just double checking
yuh its right
ok thanks
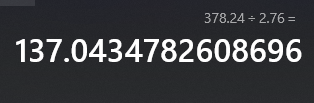
Mass: 378.24 g Density: 2.76 g/\(cm^3\) You're being asked for the volume; volume units are \(cm^3\) so we're gonna have to do a little bit of maneuvering here. First step: flip the fraction for density so the cubed centimeters are on top. Then multiply that by the mass so that the grams cancel out:\[378.24g\cdot\frac{1cm^3}{2.76g}\]After this, you should get a division expression\[\frac{378.24}{2.76}cm^3\]which should simplify to your final result. \(378.24\div2.76\approx137.04\) (see below) so your result should be around 137.04 cubic centimeters.
okie dokie
Join our real-time social learning platform and learn together with your friends!