Hello, I need help. SS below. Before we et far Idk anything abt this so help me.
what do u know about pH
Nothing.
what do u think pH is
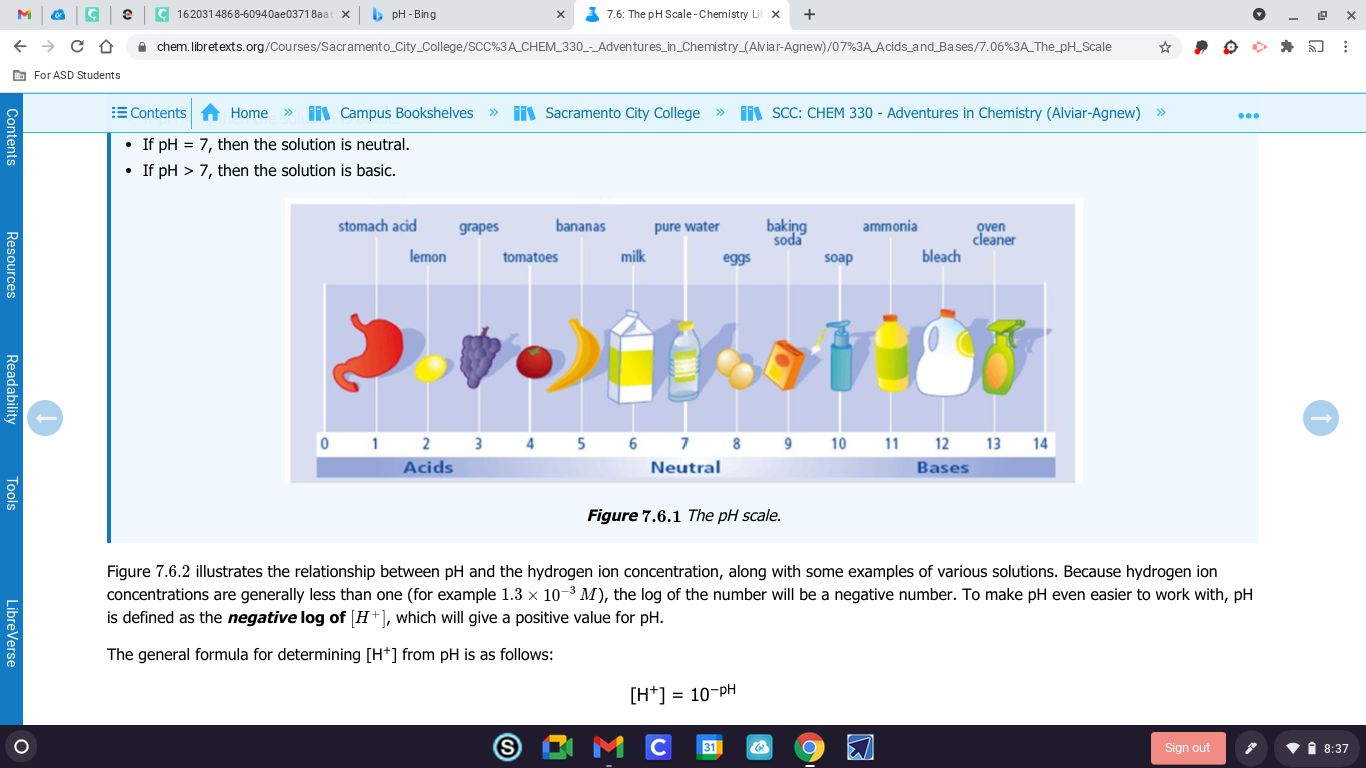
Idk but according to google it's a graph that depicts the acidity or alkalinity of a solution on a logarithmic scale, with 7 representing neutral, lower values indicating more acidity, and higher values indicating more alkalinity. The pH is equal to log10 c, where c is the moles per liter hydrogen ion concentration.
by the way look at ph>7 u just answered that by repersenting netural
I don't understand. T^T
when u look at the bottom of ur paper it says pH>7 what does 7 repersent
represents neutral? lower values indicating more acidity, and higher values indicating more alkalinity.
good job ur getting the hang of it
So I put neutral-?
yes and i have a very good screenshot to help u a bit more
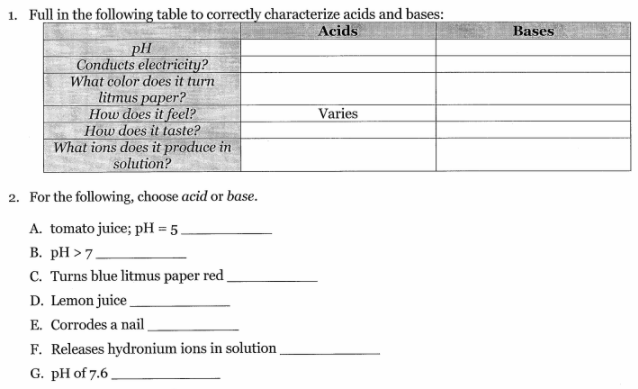
That helps but I don't think I'm supposed to put neutral. It says in my ss, "choose Acid or Base.
Shall I go with Base?
look at this screen shot ull see
here what is acid
I got #2 done. may I have help with one?
yes ill tyr my best this my first time doing this stuff tbh but im apperently good at it so tys for the oppurtunity
Yeah. My first time too. We'll try our best. ANd just in case I'll bring is snowflake if I still don't understand after a long time xD
k
so what are acids that conduct electricity
are liquids acid
Uh sorry, I was AFK. I believe the answer to conducting electricity would be Acid/yes.
that not the answer
do u know what hydrochloric is
T^T How would you know if it was the answer or not? >.> You've never done this before and- ughhh. and yes it's muriatic acid.
ok nice i am using google to btw
cool. I had a feeling you were.
I got the part finished but I've got more questions xd
ok nice good job
This is what else I need help on XD
lets try that top one
0-6 is acids right
7 is netural
8-14 are bases
Yes. 0-6 is Acid and 0-7 is Basic. (this is from my textbook >.>)
I mean 7-14. >.>
Basically: Acids: pH0-pH6 Bases: pH8-pH14 0 being strongest 6 being weakest 8 being weakest 14 being strongest
\(2\) So, to start off go through the pH levels, Using my guide figure out which ones are acids and which ones are bases, Then figure out if they are strong or weak (also using the guide)
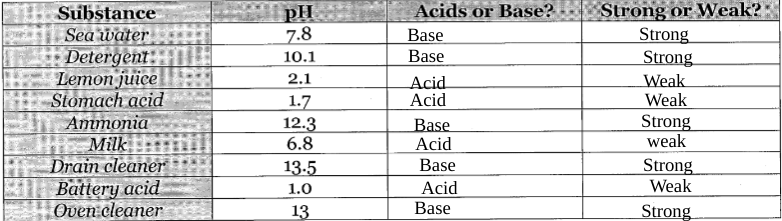
I used the information you provided and came up with this.
Make sure you specify the `strong` and `weak` into their own categories, you have acids labeled as just `strong` and bases labeled as just `weak`
strength and weakness is based upon the 2 sides, not on the whole scale
I uh I don't understand.
Yeahhhhh Ik that.
So here, I'll give you an example using `Sea water`, pH level is 7.8, so it is closer to neutral on the `Bases` side, meaning that it would be a `Base` and it would be `Weak` because it is closer to neutral than it is to pH14
ok? I'm still not catching on. I'm rlly dumb yk xd
so, look at acids and bases as seperate scales all together, 7.8 is on the low end of the scale, farthest away from 14, so it is considered weak, whereas if the number was near 14 it would be considered strong
same goes for the acids side, just reverse, 0 is the side farthest from neutral (7) and 6 is closest to neutral
yeaahhh,
you understand it now?
yeah i think so.
ok, rewrite the chart and show me when you're done
My teacher actually cleared me for the second page, She said I didn't need 2 to do it I kinda just did all of that for nun T^T
oh, well ok ig (:
Join our real-time social learning platform and learn together with your friends!