chemistry
@vocaloid
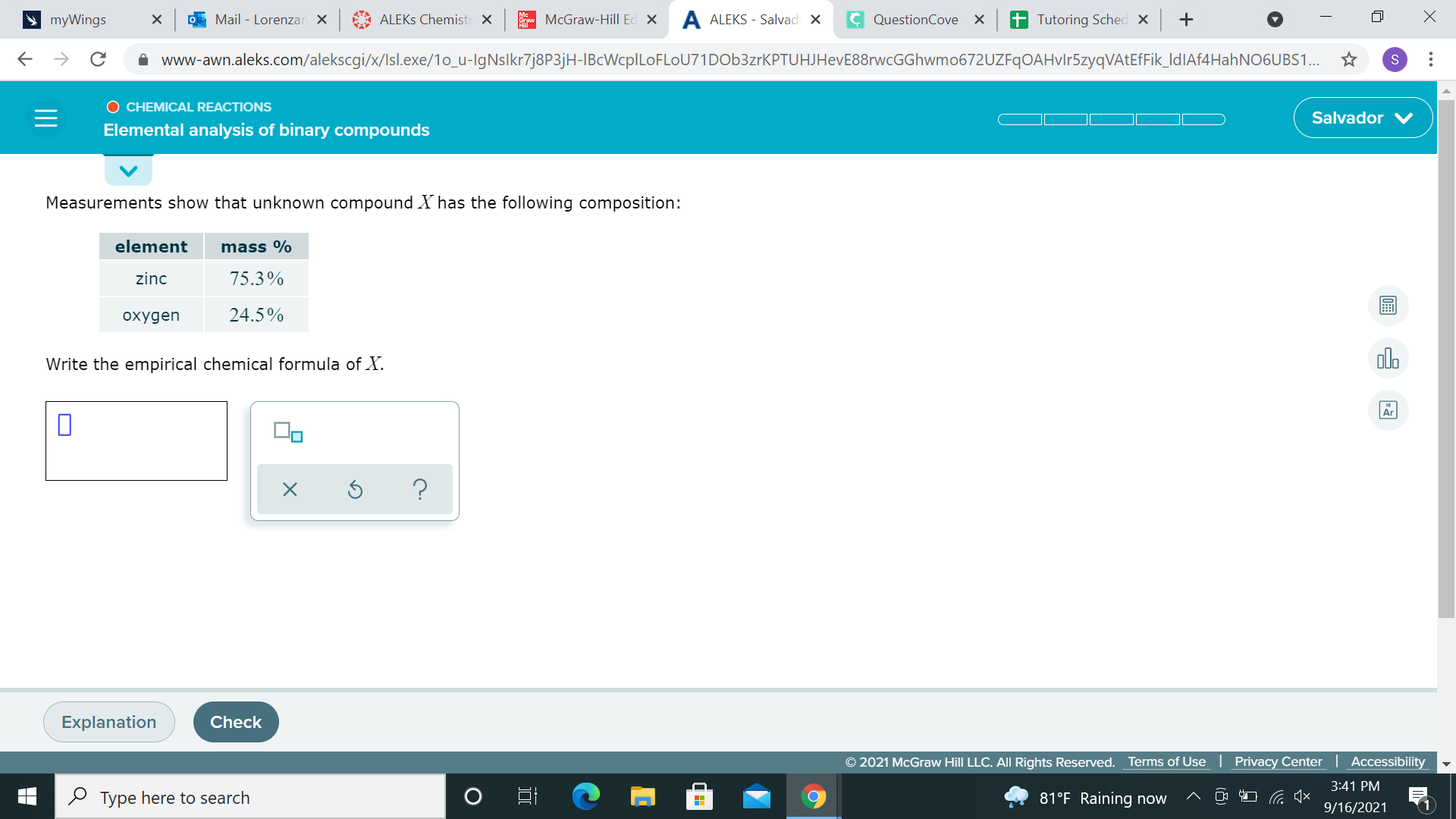
For these types of problems, assume you have 100g of the compound 75.3% zinc ---> becomes 75.3% * 100g, or 75.3g Zinc same logic with 24.5% oxygen ---> 24.5g O2 (remember, oxygen by itself exists as O2 not just O) divide each mass by the appropriate molar mass now, you'll have moles of each compound. write the formula based on the moles.
actually sorry just use O not O2 for oxygen
7530?
75.3 divided by 65.38
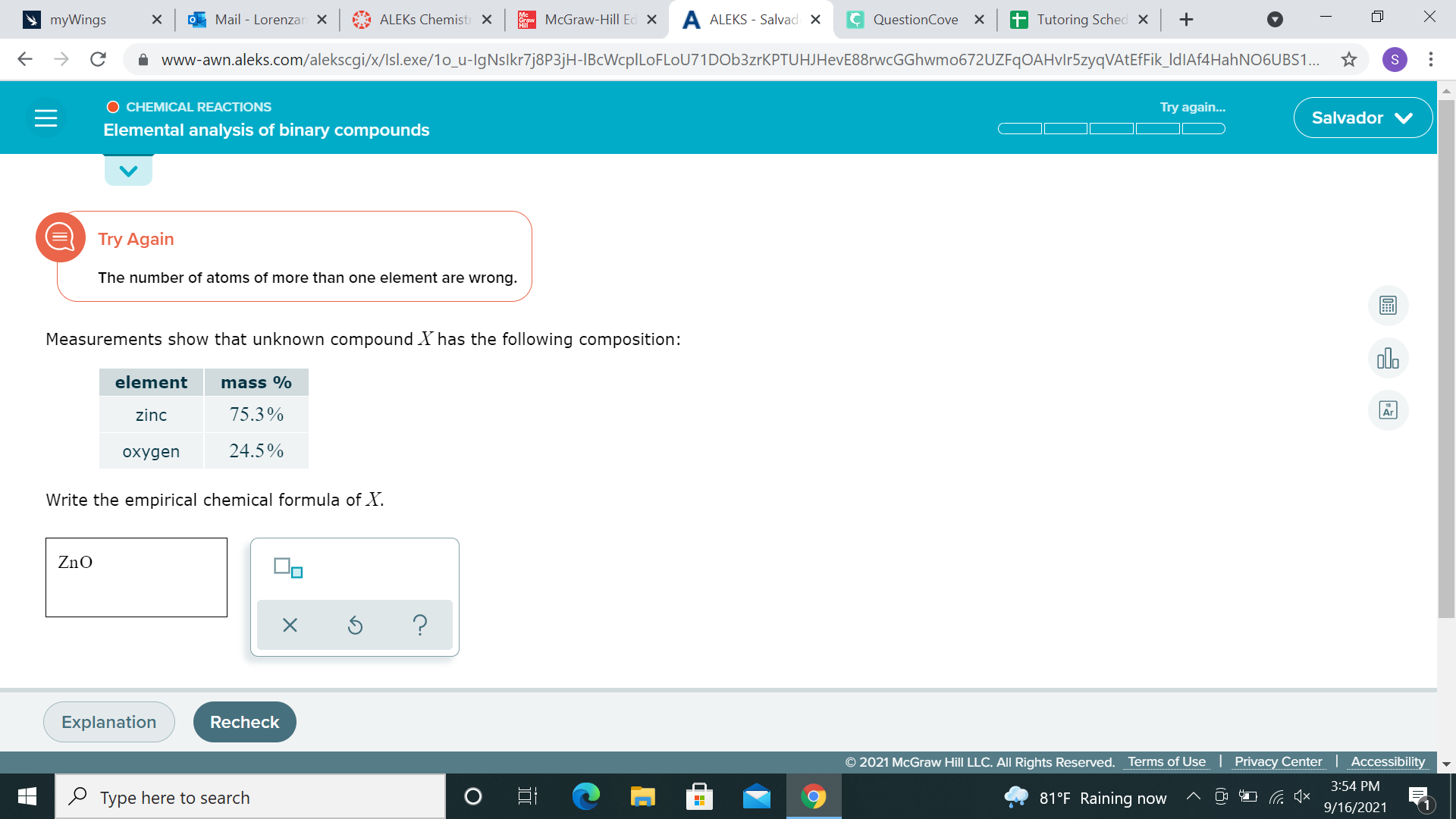
ah sorry just had to refresh myself on how to do this, I missed an important step 75.3g Zinc / 65.38g/ mol = 1.152 moles Zinc 24.5g Oxygen / 15.99g/mol = 1.531 moles Oxygen we take the lower number (1.152 moles Zinc) and divide 1.152 moles zinc / 1.152 moles zinc = 1 Zn 1.531 moles oxygen/1.15 moles zinc = 1.32 O this means our ratio is 1 Zn to 1.32 O. we can round 1.32 down to 1 to get a 1:1 ratio of Zn to O, meaning ZnO is our formula
ZnO is the answer?
yes
couldnt you have easily just connect Zn zinc and O oxygen together making ZnO without the equations?
No. Different compounds have different ratios of atoms. For example, Iron can have different charges like +3 or +2, so if you have iron oxide, you can't just put iron + oxygen together because you have different ratios of iron/oxygen based on charge.
it says its wrong
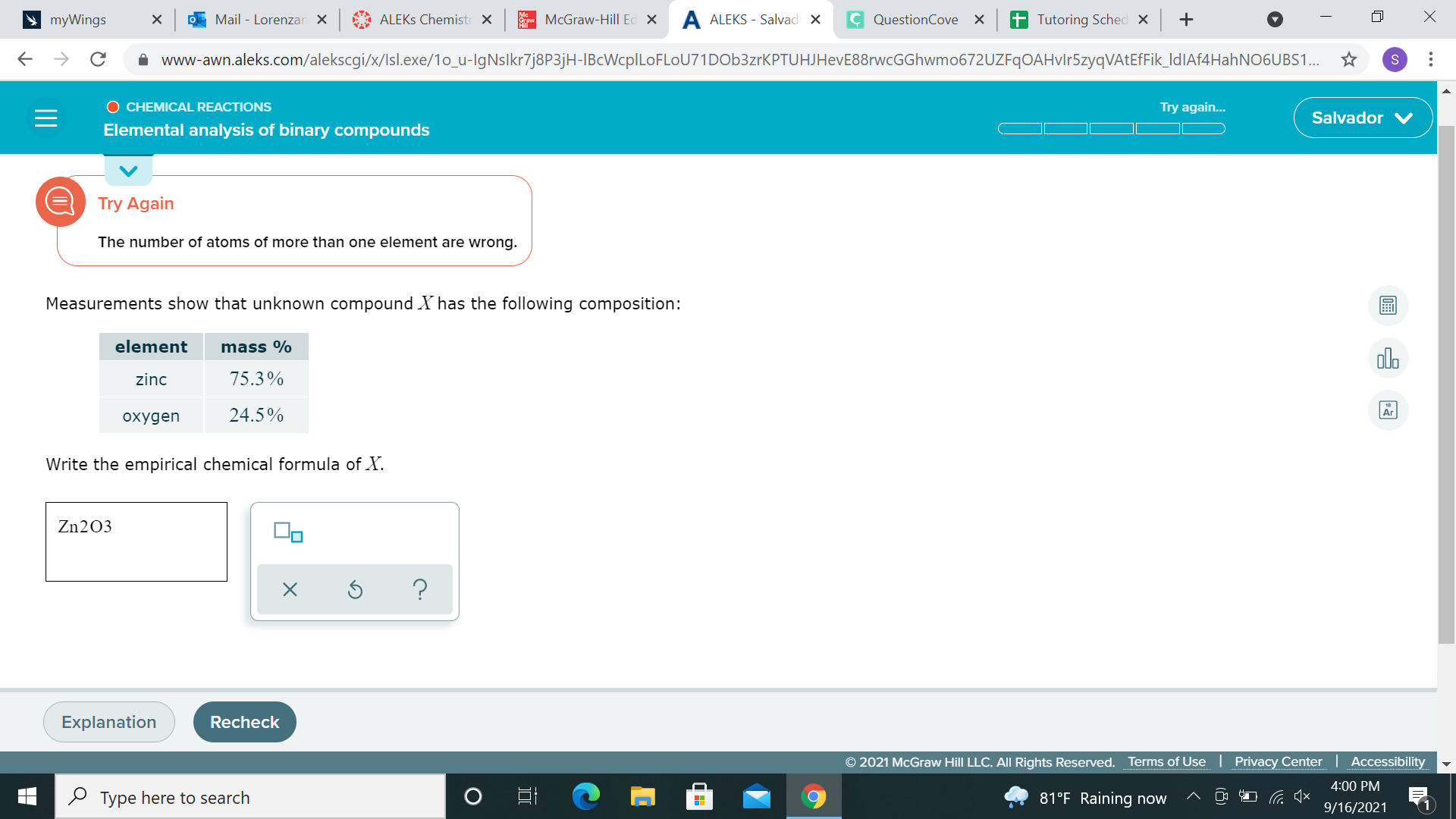
alright if your mole numbers are too far away from integers, you can multiply by whole numbers 75.3g Zinc / 65.38g/ mol = 1.152 moles Zinc 24.5g Oxygen / 15.99g/mol = 1.531 moles Oxygen we can multiply both by 2 to get 2.3 moles Zinc and 3.06 moles Oxygen, so they might want Zn2O3 instead?
Zinc is only supposed to have a +2 charge and oxygen almost always has a -2 charge, which is why I went for ZnO initially
lower 2 and 3?
yeah they need to be subscripts
the answer is Zn3O4
ok, yeah now I see it 75.3g Zinc / 65.38g/ mol = 1.152 moles Zinc 24.5g Oxygen / 15.99g/mol = 1.531 moles Oxygen 1.152 moles Zn/ 1.152 = 1 mol Zinc 1.531 moles O/1.152 = 1.329 mol O they want to multiply both by **3** to get 3 mol Z and 4 mol O
ok thanks
Join our real-time social learning platform and learn together with your friends!