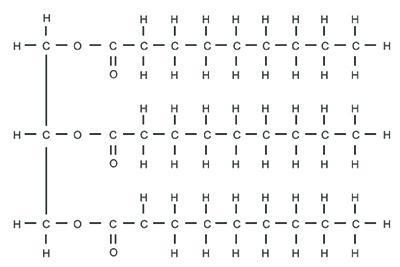
This figure shows the chemical structure of a molecule. The left side of the molecule contains three vertically oriented carbons bonded to each other. Each carbon is also bonded on its right side to an oxygen. Each oxygen is itself bonded on its right side to a long chain of carbons and hydrogens. Therefore, there are three of these long carbon-hydrogen chains within the molecule. Image Courtesy of 3DScience.com What occurs when the macromolecule pictured above mixes with water? It dissolves in water. It assumes a ring shape in water. It fails to assume a ring shape in water. It fails to dissolve in water.
Do you have any ideas for the answer?
is Chemistry a familiar topic for you?
already have the answer thanks though
ope, my apologies. Sorry you didnt get any help sooner
OP already got their solution, but just in case anyone finds this problem: the left carbons make up a glycerol, while the chains of carbons are fatty acid chains, making this molecule a lipid. This molecule is non polar due to the long hydrocarbon chains and thus will not dissolve in water.
Join our real-time social learning platform and learn together with your friends!