chemistry
@vocaloid
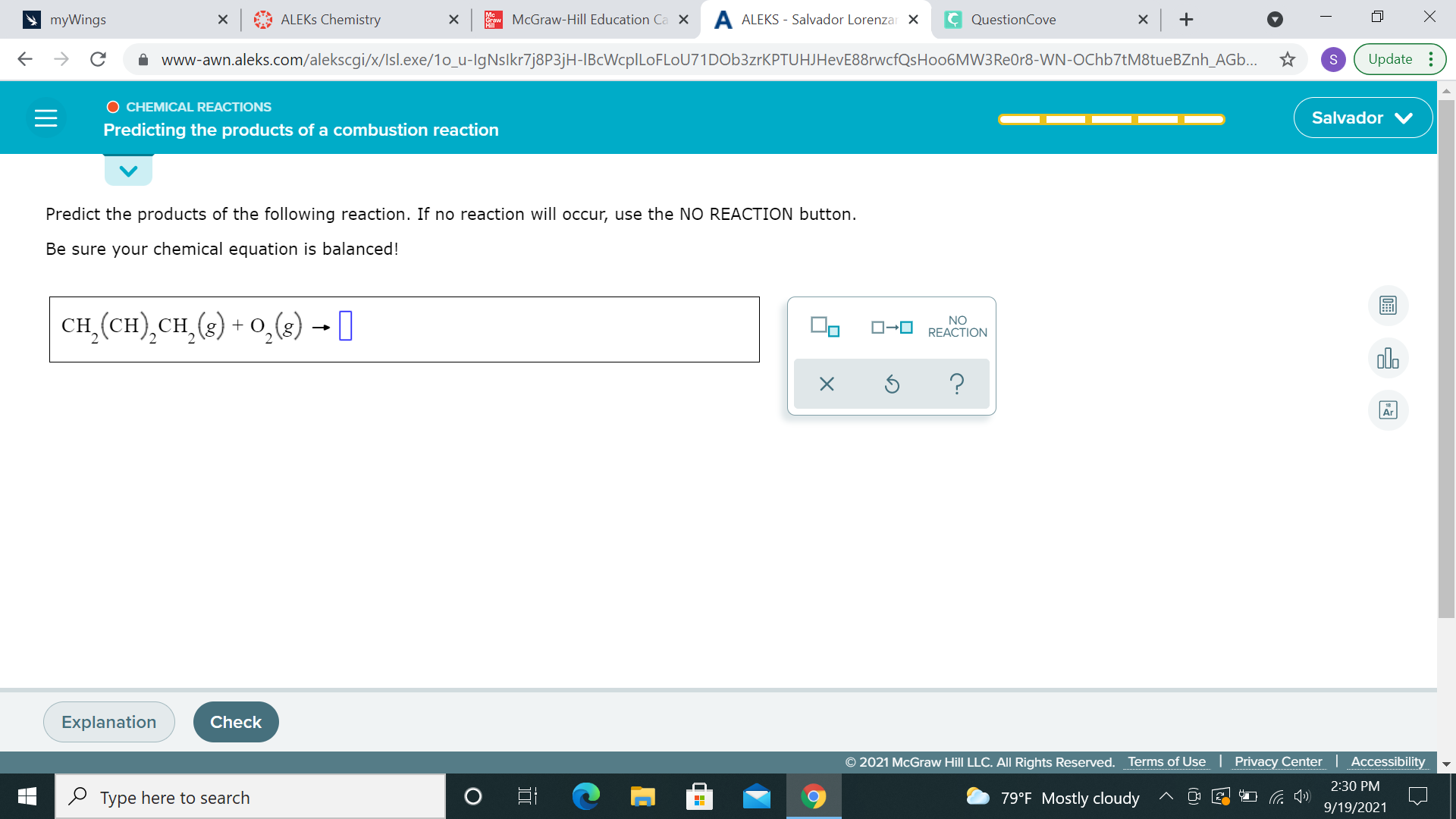
when you have a long hydrocarbon (compound w/ C-H only) + O2, this is a combustion reaction so your products are CO2 and H2O can you try balancing from there? I'll help you out if you get stuck it might be helpful if you re-write CH2(CH2)2CH2 as C4H6 since there are 4 carbon atoms and 6 hydrogen atoms
how is there 4 carbon?
how do I balance it if theres nothing to balance from
|dw:1632087387284:dw|
like I said earlier, this is a combustion reaction, so your products are CO2 and H2O CH2(CH)2 CH2 + O2 ---> CO2 + H2O, balance
i put a 3 in front of h2O
yes, that's a good place to start, now the hydrogen atoms are balanced - now try balancing either C or O
and then a 4 infront of O2 and CO2
I agree with putting a 4 in front of CO2, that way you balance out carbon however, at this point notice how you have 3H2O + 4CO2 as the products. this is 11 oxygen atoms (which is an odd number, so you can't balance this with O2), so you'll need to re-balance the oxygen atoms by increasing the products.
hint: try doubling both H2O and CO2 to get 6H2O + 8CO2
how do i balance oxygen if I cnant put a 4 infront of O
if you double both H2O and CO2 to get 6H2O + 8CO2, you have 22 oxygen atoms (so you can now balance O2 by adding an 11 in front of O2) all that's left now is to rebalance H and C by adding some number in front of CH2(CH2)CH2
so whatever number is infront of H2O multiplies all of them?
yup 1H2O = 2 hydrogen 1 oxygen 2H2O = 4 hydrogen 2 oxygen and so on
put a 2 infront of CH2?
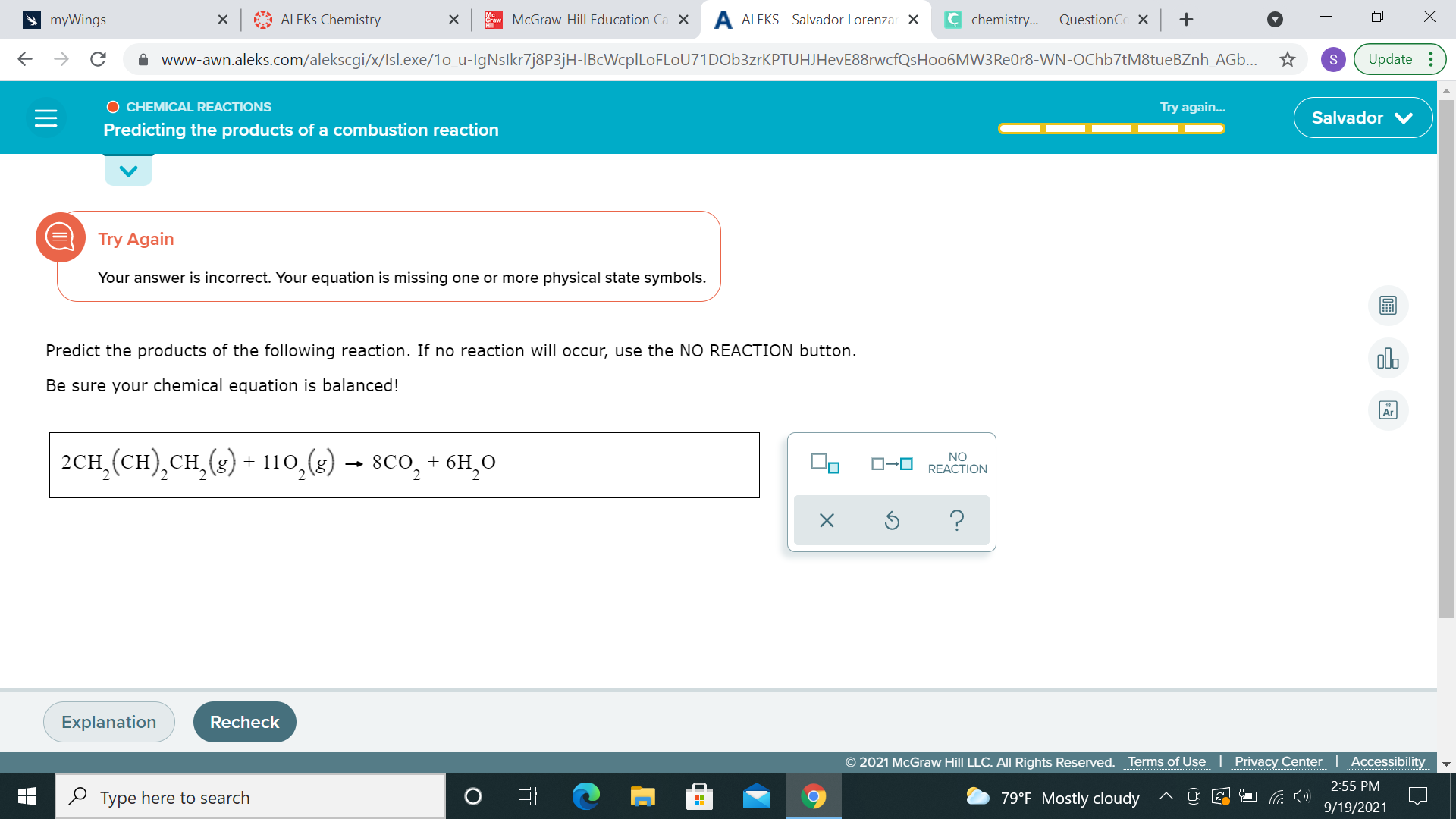
yup, 2CH2(CH)2CH2 + 11O2 --> 6H2O + 8CO2 is balanced
theres 8 H on one side and 12 hydrogen on the other side
CH2(CH)2CH2 is 6 hydrogens the first CH2 has 2 the middle (CH)2 has 2 the last CH2 has 2 then double the result to get 12
but why do i double the result
theres no paranthesis to double it
there's a 2 in front of it 2CH2(CH)2CH2 the 2 in front doubles every atom count
for combustion, H2O and CO2 are both gaseous (g)
Join our real-time social learning platform and learn together with your friends!