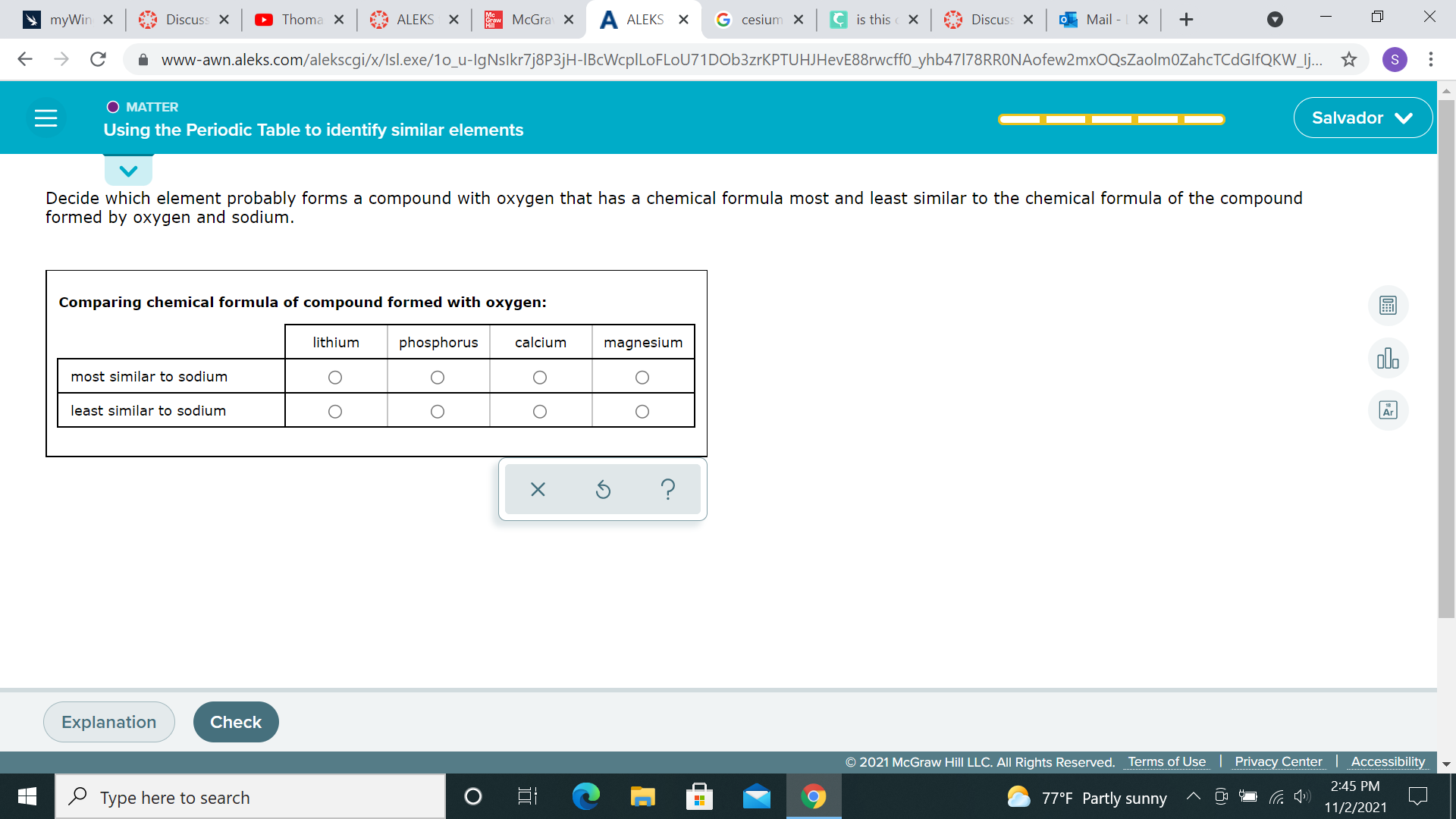
chemistry help
@nishio ur the best at this kind of stuff
@joeltheboss
@astrid1
So Lithium would be most similar to sodium because both sodium and lithium both have a electron surrounding their outermost shell. Magnesium and Calcium are close to sodium in the periodic table and are also In the same group, if I remember. So those would also be most similar to sodium. Phosphorus and sodium are different in many different ways. So therefore, it is less similar than sodium. Sorry if I get any wrong. Resources: 1. 1 2. https://www.schoolmykids.com/learn/interactive-periodic-table/compare-sodium-phosphorus 3. https://www.schoolmykids.com/learn/interactive-periodic-table/compare-lithium-sodium
joeltheboss is he correct?
also I can only pick one for the most similar and only one for the least similar
@astrid1 I can only pick one for the most similar and one for the least similar
What do you mean?
your telling my multipule of them are the most similar and multipule of them are the least similar when it can only be one for each
@mathplayer223 from what he wrote, Lithium would be the most similar and Phosphorus would be the least similar
okay thanks guys
Thanks Astrid
Join our real-time social learning platform and learn together with your friends!