help last question
@vocaloid I'm not really sure where I get the daughter nucleus part. I know how to do decays.
For each step look at the atomic number and atomic mass. There’s 7 steps where you have one daughter nucleus and 2 steps at the end where you have two nuclei each First step: atomic number 91 atomic mass 142 This is proctactinium 142 91 Pa (On my phone but you get the formatting)
So, I am confused. Will I need to find alpha and beta for all the steps or just identify the decays on the graph?
@vocaloid
It's just asking for daughter nuclei, so no alpha or beta particles
okay, so will I have 7 answers then?
hm... I'm looking at the diagram again, I think there are actually 11 daughter nuclei (I'm not sure I entirely understand the last decay step myself)
sorry I'm going to get a second opinion on this @AZ would you mind taking a look at this, I'm not 100% clear on this diagram
okay, thank you! I am trying where the answer splits and goes horizontal so like 9 and see if I am correct. I have a few attempts on this.
It's really hard to read the image but is the question asking you to list every single daughter product formed?
\[\begin{matrix}\text{A} \\ \text{Z}\end{matrix} X\] Z is the atomic number which is just the number of protons A is the mass number which is number of protons + neutrons
Your graph tells you the number of neutrons on the y-axis and the number of protons on the x-axis You have to use the number of protons to find the element on the periodic table You then add the number of protons and neutrons to get A which is the mass number X is going to be the element from the periodic table that corresponds with the number of protons
Remember: if the number of protons changes, then you get a completely different element if the number of neutrons changes, you have the same element but with different masses and they're called isotopes. If the number of electrons changes, you form ions and they can be either cations or anions depending on if you lost an electron or gained an electron respectively.
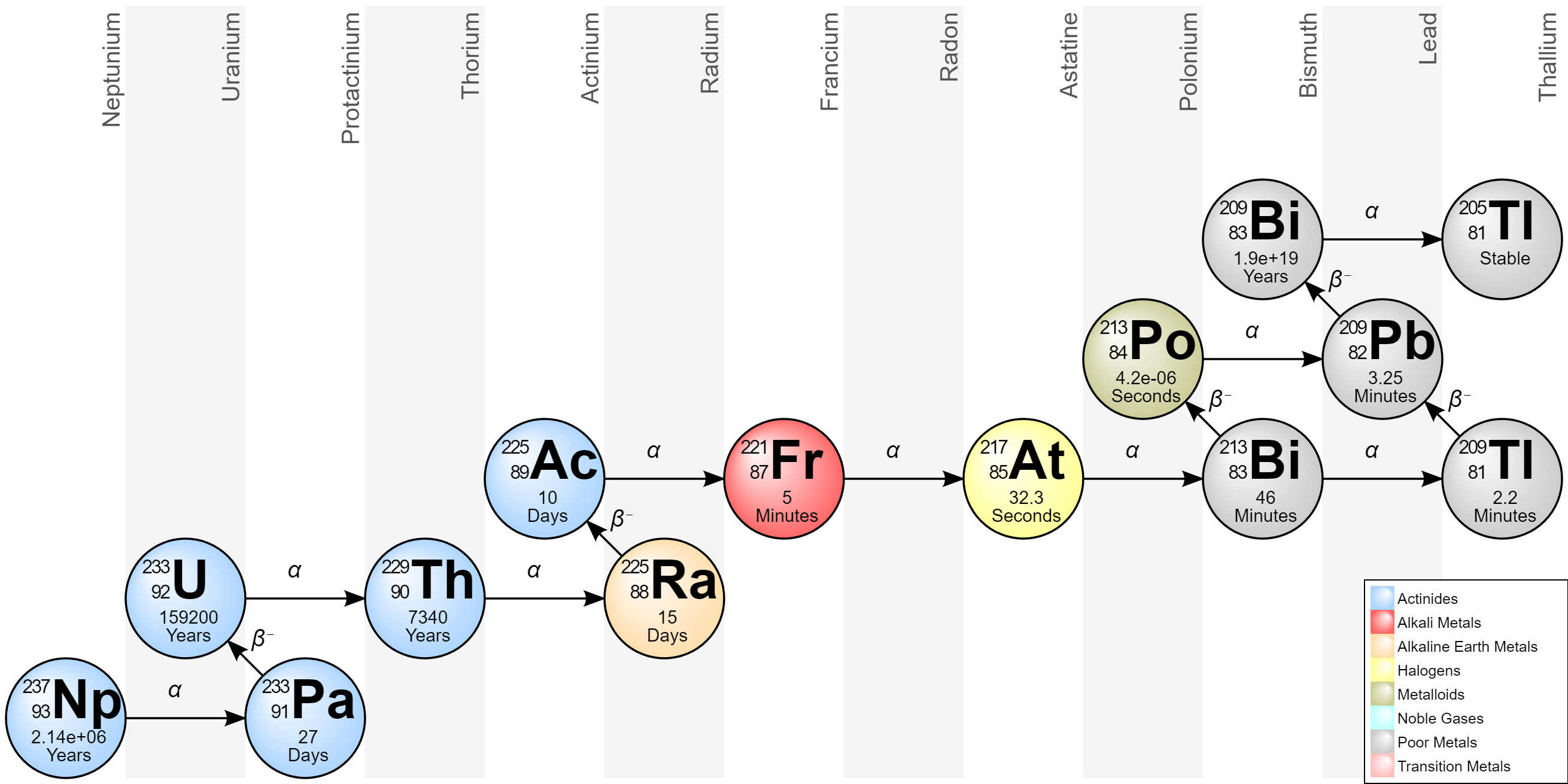
Anyway, I found a better image of neptunium-237 decay that might be easier to read and help you list them out much faster The only exception is that this diagram lists titanium-205 which isn't in your figure
You won't even have to add the protons and neutrons from your figure since this image gives you it all. It even lists what sort of reaction it is (alpha or beta decay).
Join our real-time social learning platform and learn together with your friends!