Can someone help me please: How many total valence electrons, total domains, total bonded atoms, and lone pairs for compound SiH4
I already know that there are 8 valence electrons and maybe 0 lone pairs, but i need to figure out how many total domains there are and how many total bonded atoms
Can you draw the structure first?
Good! How many electron pairs are there? A pair of electrons = 2 electrons And of those pairs, how many of them are bonded and how many are a lone pair?
There are 4 sets of 2 electrons
Are they all bonded pairs?
Yes i believe they are
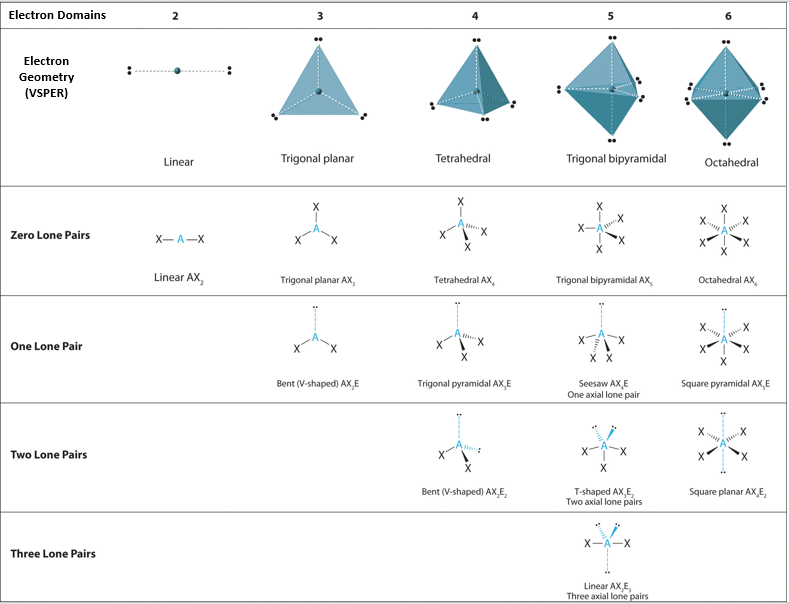
The total domains refers to how many pairs of electrons there are and includes bonded pairs as well as non-bonded pairs of electrons. total bonded atoms refers to how many atoms (that is hydrogen in this question) are bonded to the central atom (which is silicon).
Can you answer your question now?
So i feel like there are 4 total bonded atoms
And i believe that there are 0 lone pairs
So with saying that there are 4 total domains am i correct?
Correct! All the valence electrons are bonded and thus there are no lone pairs.
Good job!
Thanks you for the help
It was my pleasure!
Wait what is the molecular structure (or shape) of SiH4 and also the VSEPR (or electron geometry)
When there are no lone pairs, the molecular structure is the same as the electron geometry. If there are lone pairs, then the molecular structure depends on how many lone pairs. We have 4 electron domains so what would the shape of SiH4 be?
AX4 just stands for the type of molecule such as SiH4 or CH4 Tetrahedral is correct
Ok thanks
You're welcome!
Join our real-time social learning platform and learn together with your friends!