What is the electronegativity difference for the compound NH3, is the compound symmetrical or asymmetrical, and is the compound polar, nonpolar, or ionic?
Hello. This question is composed of three parts
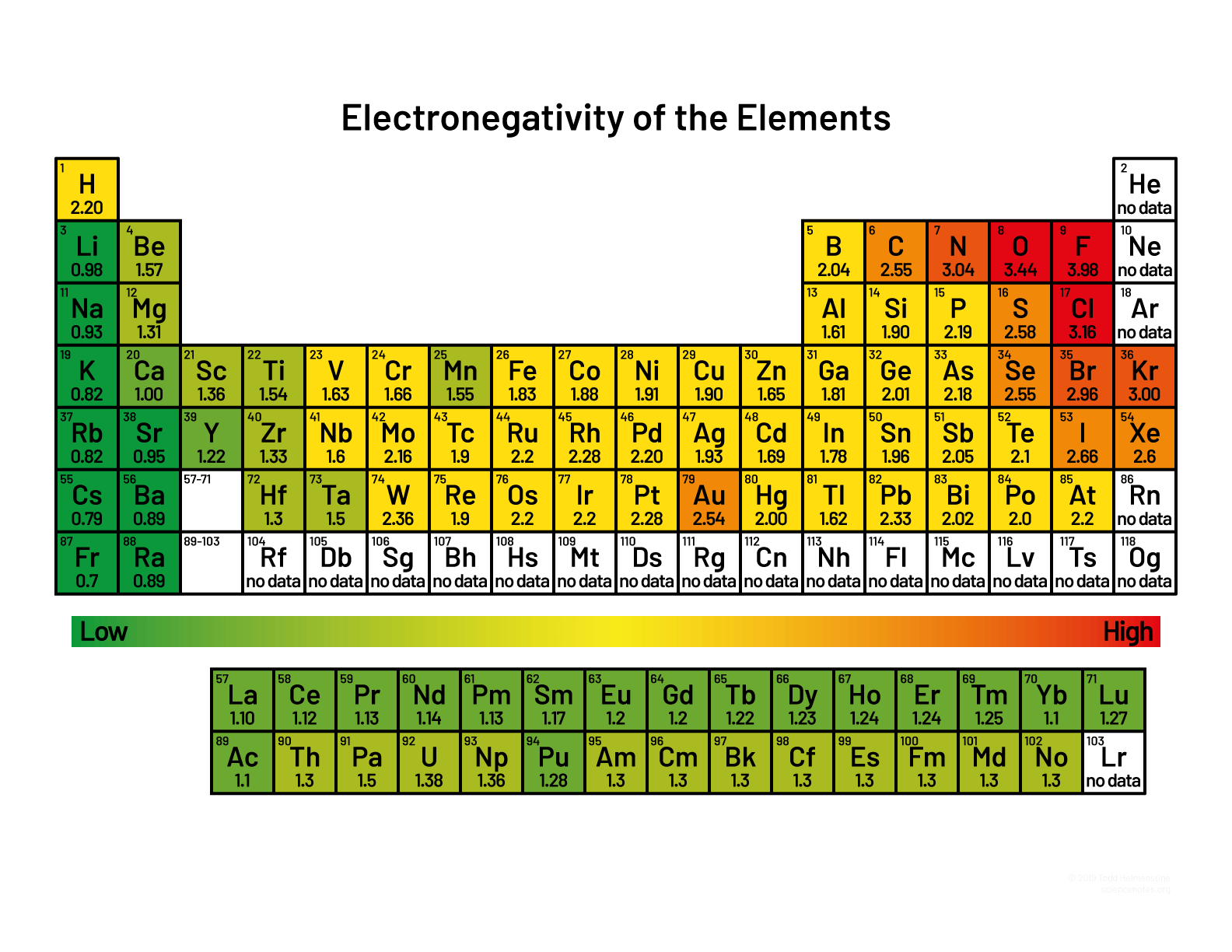
a) Electronegativity Difference I'm hoping this means the individual electronegativities between the 2 N-H bonds. If not, don't listen to me lol. Your Lewis dot structure is very much correct, and to take it a step further the compound shape will be a trigonal pyramid. When finding the electronegativity difference, consult this picture here. You should be able to find the two individual atoms, Nitrogen and Hydrogen. You can find the difference by subtracting the bigger minus the smaller electronegative number.
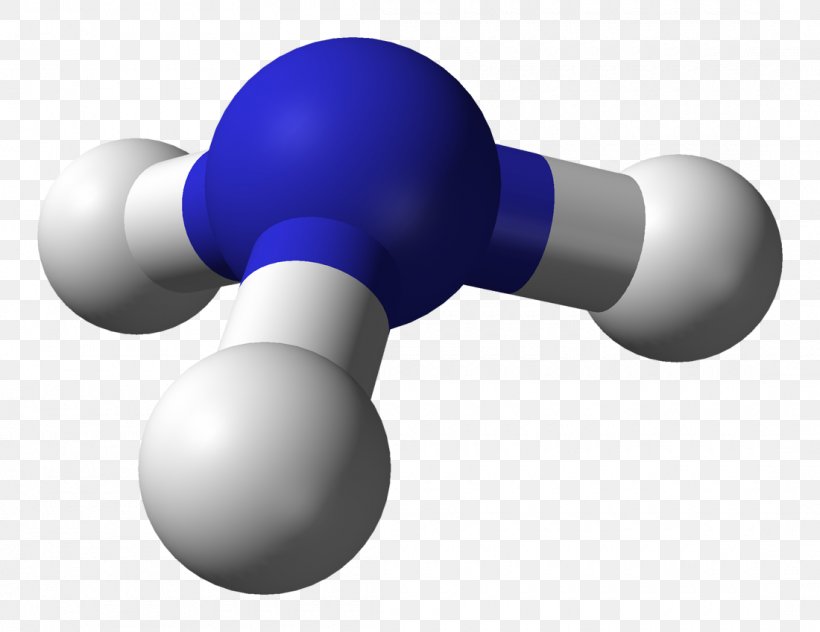
b) Symmetry Imagine the compound NH3 as a marshmellow as the atoms and the sticks representing the bond. (imagine lone pairs as an invisible marshmellow bond for now.) Evenly distribute those, and it should look something like this. To determine the symmetry, look at the axis and center of symmetry. If it has both, it is symmetric. if it does not, it is assymetric.
c) Polar/Nonpolar If the answer you got before is asymmetric, the molecule is polar. if you said it was symmetric. it is nonpolar.
Hope this helped.
Yes. Should be correct
Yes!! Totally correct. And with that answer you should be able to find the polarity and non polarity decision
Which means the molecule is polar
Yep!
Thanks for the help!!!
Join our real-time social learning platform and learn together with your friends!