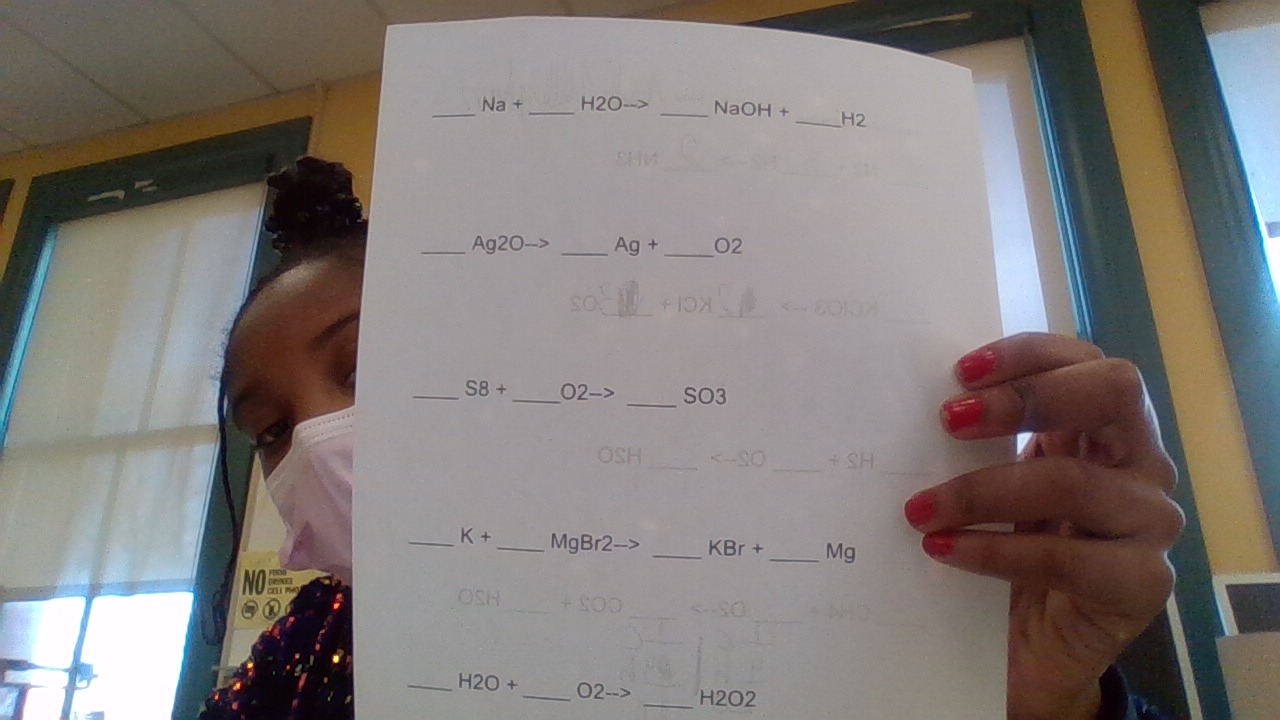
h3lppppp
what are we supposed to put
Please keep in mind this is an educational website. We can help but we will not do the whole assignment for you. For these problems, you want to balance out the equations by making sure both sides have equal numbers of atoms. You can do this by adding numbers in front of the compounds to change the numbers of atoms on each side. So for the first one: _Na + _H2O —> _NaOH + _H2 Notice how you have 2 H atoms on the left side, but 3 on the right. So you can write a 2 in front of H2O and a 2 in front of NaOH to get _Na + 2H2O —> 2NaOH + _H2 Now you have 4H on each side. However, the Na atoms are now unbalanced, so you’ll re balance those using similar logic. Keep adjusting until all compounds are balanced.
Join our real-time social learning platform and learn together with your friends!