Determine the mass in grams of 4.54 ✕ 1024 molecules of CO2. @vocaloid
10^24
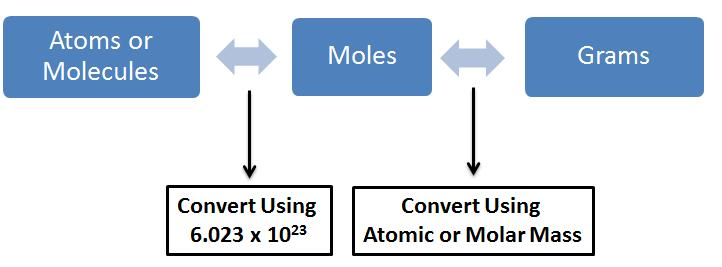
Divide by Avogadro's constant to convert it into moles and then multiply by the molar mass of CO2 which is 44.01 to convert it into grams
You should keep this flow chart in your mind when you're converting between molecules/atoms, moles, and grams
4.54 * 10^24 molecules Avogadro's number is 6.023*10^23 First divide to get the number of moles: (4.54*10^24) / ((6.023*10^23) = ??? and then multiply by the molar mass which is 44.01 to convert it into grams of CO2
7.54
I haven't done any calculations yet but you should include units. Is that 7.54 moles? Or 7.54 grams?
Moles I did the equation you had up
I wouldn't round just yet so that way you can get the most accurate answer But the next and final step is to multiply by 44.01 grams / 1 mole to convert from moles to grams
so take what I have in my calculator and multiply the grams/1 (or the number I got for moles)?
what you have in your calculator, you multiply it by 44.01
3.17373 with just multiplying
Check again 7.5377 * 44.01 = ?
331.73
The molar mass tells us how many grams of that compound is in 1 mole So we multiply it by the number of moles to find out how many grams are in that many moles
thank you
Join our real-time social learning platform and learn together with your friends!