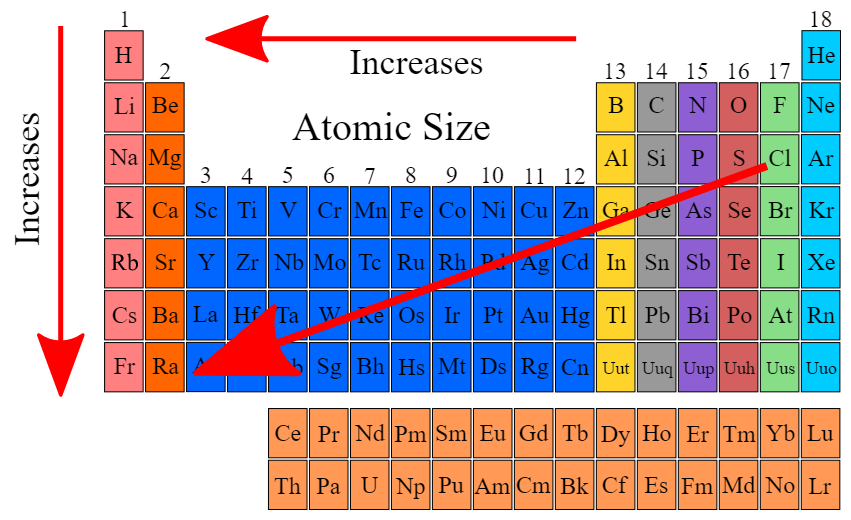
Sulfur is to the immediate right of phosphorus in the same row in the periodic table. Which of the following statements compares the radius and number of protons of nitrogen and oxygen atoms? Sulfur has a smaller radius and one more proton than phosphorus. Sulfur has a smaller radius and one proton less than phosphorus. Sulfur has a larger radius and one proton less than phosphorus. Sulfur has a larger radius and one proton more than phosphorus.
Am I right with A?
@tetsxpreme
@axie
I believe so, excuse me if im wrong, but my strive subject isn't science, but yes indeed i think you are correct
forgive me if i am wrong
So yes, indeed you are correct.
Atomic radius increases when you go down a row or towards the left on the periodic table
So it goes without saying that it would decrease if you went towards the right which sulfur is to the immediate right of phosphorus in the same row
That is correct
thank you
You're welcome
the worst mod ever lmao
Join our real-time social learning platform and learn together with your friends!