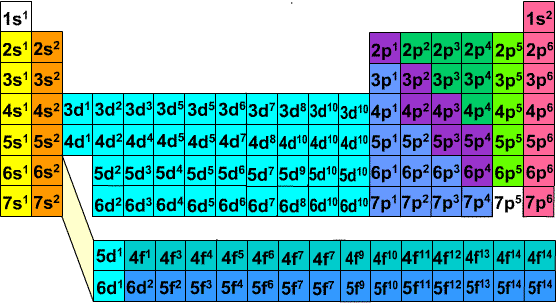
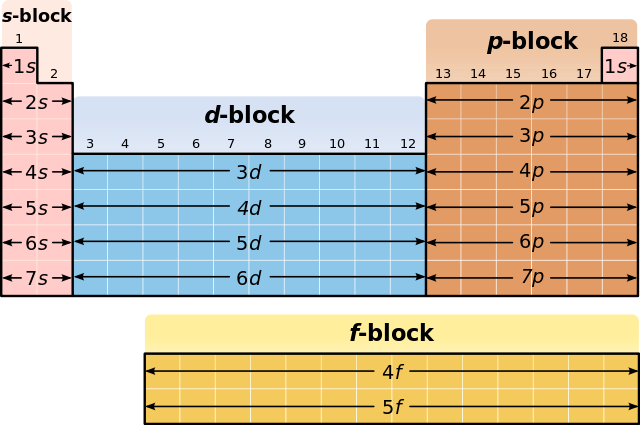
The electron configuration of an element is shown below. 1s22s22p5 Name the group this element belongs to in the periodic table and explain your answer. Based on the electron configuration, explain how many electrons it is likely to receive from the atom of another element during the formation of a bond.
what do you think it is
Any ideas?
What element would 1s2 2s2 2p5 be? Here are some example of other elements and their electron configuration
This element belongs to the halogen group and the element is also fluorine.
@tranquility
You found it by looking at the periodic table
Because fluorine is the only element with that electron configuration
The first image I attached clearly tells you which orbital the last electron is in. If you don't know the s, p, d, f blocks then this should help
Yes
Ok so how does 1s22s22p5 belong to the halogen group
As for the second half of your question: `Based on the electron configuration, explain how many electrons it is likely to receive from the atom of another element during the formation of a bond.` How many electrons does it need for it to have the same electron configuration of a noble gas? Remember, halogens are very reactive. Think of HI, HCl, HF How many electrons is the hydrogen sharing with these halogens?
1?
Yes
Ok thank you so much <3!
You're welcome!
Join our real-time social learning platform and learn together with your friends!