More chem please help: https://snipboard.io/bU8BrY.jpg
..
The link don't work?
@tranquility
nope
Are those the only three answer choices
Yes indeed
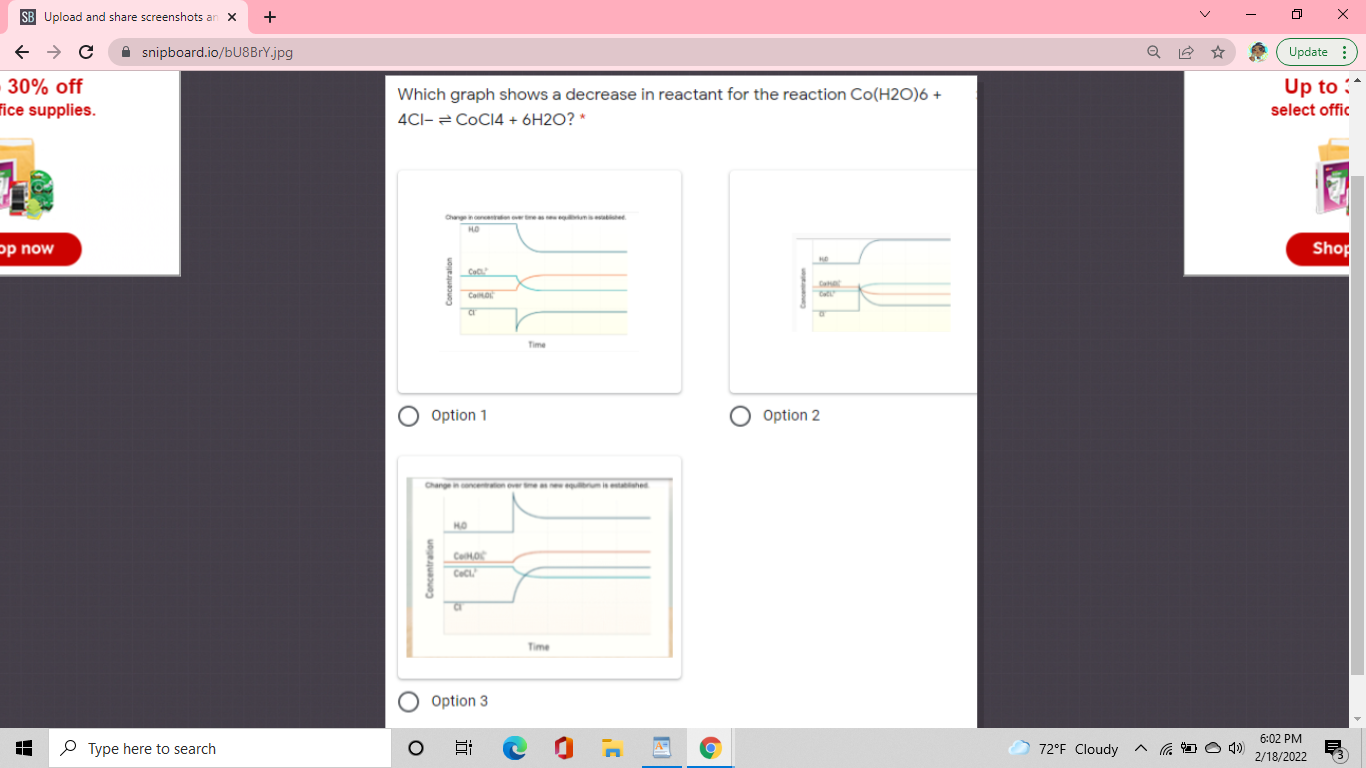
The equation is Co(H2O)6 + 4Cl- <----> CoCl4 + 6 H2O We have two reactants Co(H2O)6 and Cl- We want to see a decrease in either Co(H2O)6 OR in Cl- The way we see decreases are by the sharp straight line and it has to go downwards to signify that it is decreasing
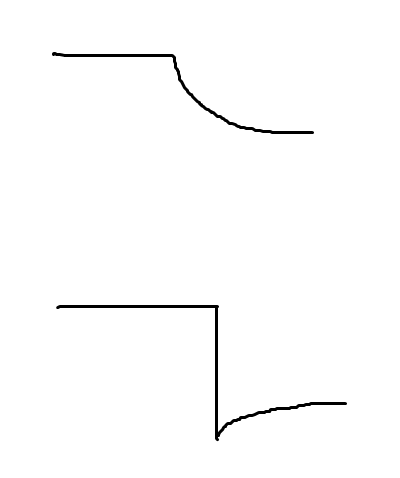
So I drew out the two possible ways we see a decrease The first one tells us that the other reactant was added into the reaction and so the equilibrium shifts towards the other side and that's why whatever compound it is is being used up to form more products. The bottom one tells us that something was removed and thus the equilibrium shifted towards it. So if you remove a reactant, then it's concentration would decrease sharply (thus the straight line) but now the equilibrium is disturbed and it's going to start forming more of it which is why the line starts to go back up slowly
Does that make sense?
Ohhhh i see
Basically, when the equation is in equilibrium all four line should just be a straight horizontal line
But then when you decrease the reactant, the line of that reactant will sharply decrease first and then the equilibrium will shift towards it and cause the reactant to increase again in concentration
Alright so with this information it sounds like its going to be option 1
So in that drawing I drew, you want to find the answer choice that looks like the bottom one for either Co(H2O)6 or for Cl-
Ohhhhh okkkk i get it nowww
That's great! We can move on to your next question now
Join our real-time social learning platform and learn together with your friends!