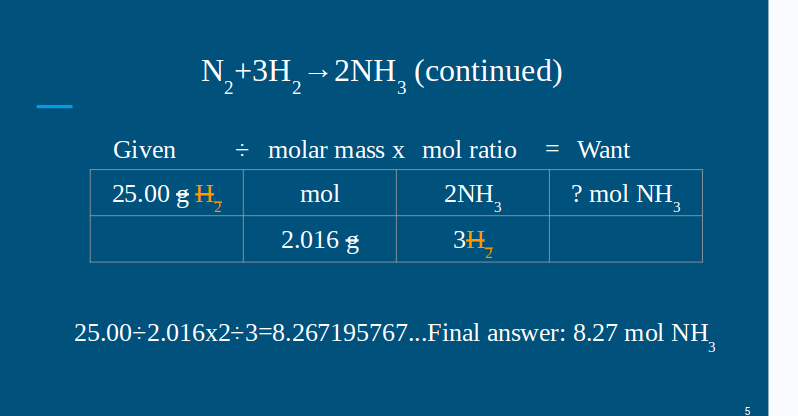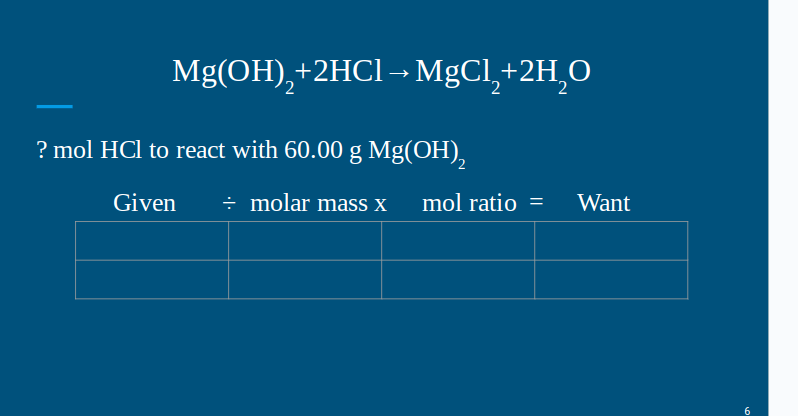HELP grade checks today and im failing i dont wanna be kicked off the team 1- Using the balanced equation on slide #4 or #5, how many moles of hydrogen gas are needed to produce 50.00 g of ammonia? Be sure to round off the the hundredths place (nearest penny, 1c)- NO UNITS, NUMBER ONLY 2- Using the balanced equation on slide #6 or #7, how many mol water are produced if 40.00 g HCl are reacted? Be sure to round off the the hundredths place (nearest penny, 1c)- NO UNITS, NUMBER ONLY 3- Using the balanced equation on slide #6 or #7, how many mol magnesium hydroxide will react with 60.00 g HCl? Be sure to round off the the hundredths place (nearest penny, 1c)- NO UNITS, NUMBER ONLY 4- Using the balanced equation on slide #6 or #7, how many mol water will be produced if 100.00 g of magnesium chloride was also produced? Be sure to round off the the hundredths place (nearest penny, 1c)- NO UNITS, NUMBER ONLY
Can you add pictures?
the balanced equation is- Given ÷ molar mass x mol ratio = Want
Oh god, I hate chemistry. I'll try my hardest to help you.
okay thank you
yeah anytime. might take me a few minutes. Is that Okay?
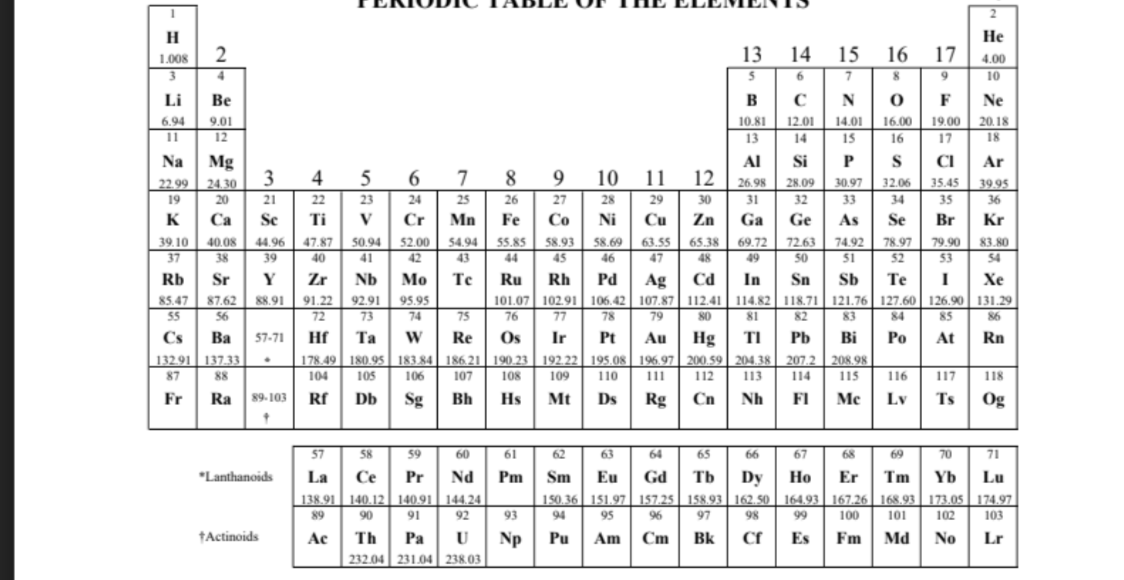
also theres the periodic table
Thanks
thank you so much!!
Yep.
I'm sorry I can't get any farther. I don't know much about chemistry, and also I didn't pay any attention either. If I knew more wI would help but ik nada.
Join our real-time social learning platform and learn together with your friends!